Whilst consultants fantasize about Pharma 4.0 and wonder when AI will replace humans, most pharmaceutical manufacturing teams rely on paper batch records — partially or entirely. For a reason: implementation of MES with integrated sensors and data is a costly and lengthy transformation process.
However, the drawbacks of paper-based processes are significant: repetitive basic checks, poor collaboration functionalities, no possibility for simultaneous work, zero data for analysis, no reusable data for other documents. This leads to lengthy reviews, delayed error spotting, extended cycle times, and bored teams who miss errors.
In 2026, digitalisation and automation are no longer an all-or-nothing, multi-million dollar question. It is a large, diverse landscape of solutions that bring different combinations of digitalisation, automation, and required investment.
In this article we outline the different options and our own view on the investment/reward trade-offs.

The batch record digitalisation landscape: from document management to fully integrated EBR.
How to Evaluate the Different Digital Batch Review Solutions
Not all digital solutions address batch review challenges in the same way. To compare them effectively, pharmaceutical companies should evaluate each option across two key dimensions.
1 — Implementation Complexity & Costs
- Organisational & integration impact on current processes
- Total cost of ownership: licences, validation, maintenance
- Time to value: how quickly measurable gains appear
- Scalability across sites and products
2 — Efficiency Gains & Quality Impact
- Impact on batch review speed and release cycles
- Impact on deviation detection speed and trends
- Gathering of process analytical and manufacturing data
- Team motivation and satisfaction at work
⚙️ Complexity vs Impact Framework
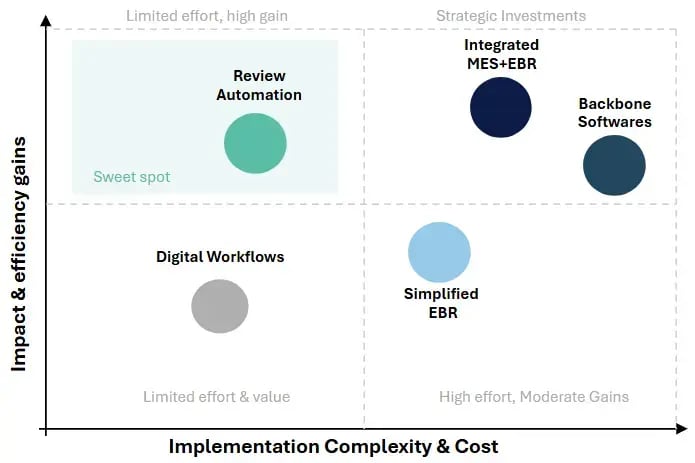
Low complexity, low impact
Fast to deploy, but limited automation value
High complexity, moderate impact
High effort for incremental review improvements
High complexity, high impact
Transformational but resource-intensive
Low complexity, high impact ⭐
The "sweet spot" for fast, measurable ROI
💡 Key Insight
Machine-learning-powered document automation delivers high impact with relatively low complexity by targeting batch review bottlenecks directly — without disrupting existing MES or shop-floor operations.
Integrated Electronic Batch Records: The Gold Standard
What EBR Solutions Address
EBR systems replace paper batch records with structured, digital workflows at the shop-floor level. They guide operators through manufacturing processes in real time while capturing data electronically, improving execution accuracy and reducing transcription errors.
Implementation Reality
EBR deployments are powerful but require substantial commitment:
- Deep integration with MES, automation systems, and ERP platforms
- Extensive process redesign: workflows must be digitalised and standardised
- Significant validation and training: operators and reviewers need new skills and procedures
- For full data integration: retrofitting of machines with sensors, where possible
As a result, EBR projects deliver long-term value but require considerable upfront investment and multi-year implementation timelines.
✅ Pros
- Eliminates paper at the source, improving data integrity
- Improves manufacturing accuracy and right-first-time execution
- Delivers strong long-term efficiency gains across the manufacturing lifecycle
⚠️ Cons
- High upfront investment in software, integration, and validation
- Long deployment timelines, often 18–36 months for full sites
- Less suitable for organisations seeking fast, incremental improvements
Leading EBR Vendors
- Werum PAS-X – Industry-leading MES with strong EBR capabilities
- MasterControl – Comprehensive eQMS & MES with strong document and training management
- SAP Digital Manufacturing – Integrated with SAP ecosystem for end-to-end visibility
- Siemens Opcenter Execution Pharma – Comprehensive manufacturing execution for pharma
- Emerson DeltaV / Syncade – Process automation with built-in batch management
- ETQ Reliance – Flexible quality management platform with manufacturing integration
- Leucine – Cloud-native EBR platform designed for agile deployment
Batch Record Review Automation: The Fast ROI Solution
What Makes This Category Different
Machine-learning-based batch record automation focuses on streamlining the review process without replacing existing manufacturing execution systems. This targeted approach addresses the primary bottleneck while enabling further digitalisation steps, such as paper-on-glass or EBR adoption. These solutions typically:
- Ingest existing batch records (PDF, scanned images)
- Extract and validate critical data automatically using ML or LLMs
- Flag discrepancies, missing entries, and deviations based on learned patterns
- Enable focus on exceptions instead of repetitive, page-by-page checks
Why ML Document Automation Delivers Faster ROI
- Minimal disruption to shop-floor operations
- No changes to manufacturing execution required
- Improved consistency and audit readiness
- Standardised checks reduce variability and human error
This combination of high impact and lower complexity makes ML automation an attractive entry point for batch review digitalisation.
Leading Solutions
- Acodis – Automation of document reviews and compliance checks, specialised in pharma batch records, quality and regulatory documents
- Aizon – AI-powered platform that optimises pharmaceutical manufacturing with real-time, GxP-compliant predictive analytics
- Mareana – AI-powered platform for manufacturing intelligence, including workflow and document automation
- Ad-hoc – Deployment by agentic solution providers such as Tulip or ThinkTrends
✅ Pros
- High impact on batch review speed with measurable time savings
- Lower implementation complexity than EBR or MES replacements
- Faster time to value, often within 6–12 months
- Scales well across multiple sites without heavy integration
⚠️ Cons
- Does not replace MES functionality at the manufacturing execution layer
- Performance depends on document clarity and quality
- Requires configuration and adaptation to company-specific rules
See batch record review automation in action
Get a personalised demo and free digitalisation audit — no commitment required.
Book a free audit →Digital Document Management: The Workflow Support
Role in Batch Review
Document management systems provide a digital foundation by enabling centralised storage, version control, audit trails, and electronic review and approval workflows. They are often the first step in the digitalisation journey, replacing physical archives with structured digital repositories.
Limitations for Batch Automation
While valuable for compliance and organisation, document management systems do not fundamentally change how batch review is performed. Manual checks remain the dominant activity — reviewers still read through records page by page, just on a screen instead of paper.
Key Providers
- AmpleLogic – Purpose-built for regulated document control
- Informetric – Pharmaceutical-focused document and knowledge management
- Kivo – Quality document management for life sciences
- OpenText Documentum – Enterprise content management with life sciences capabilities
✅ Pros
- Improved document control, searchability, and traceability
- Easier compliance management with centralised audit trails
- Relatively lower cost and faster deployment
⚠️ Cons
- Limited automation intelligence or decision support
- Modest impact on batch release timelines
- Does not reduce reviewer workload significantly
Backbone Manufacturing & Quality Software: The Broader Transformation
What These Systems Do Well
Enterprise backbone platforms provide a comprehensive foundation for quality and manufacturing operations. They typically support:
- Electronic quality management (documents, deviations, CAPA, training)
- Integration with manufacturing and laboratory systems
- End-to-end traceability and audit readiness
These platforms are often central to long-term digital transformation strategies.
Trade-Offs
Despite their strengths, backbone systems come with significant challenges:
- Long implementation timelines, often spanning years
- High validation and change management effort
- Substantial licence and integration costs
- Broad scope, even when batch review is the primary pain point
Top Providers
- Veeva
- MasterControl
- TrackWise (Honeywell)
- ETQ Reliance
- TetraScience (focus on data rather than documents)
✅ Pros
- Strong compliance backbone
- Suitable for global, multi-site organisations
- Robust audit readiness
- Modularity: possibility to combine elements for tailored solutions
⚠️ Cons
- High cost and complexity
- Slow time to value for batch review improvements
- Often over-scoped for targeted automation initiatives
Side-by-Side Overview of Solution Categories

⚙️ When Each Category Makes Sense
- Smaller or mid-sized organisations benefit most from ML-powered review automation as a starting point
- Existing sites gain the fastest ROI by layering automation on top of existing processes
- New facilities can justify full EBR and MES deployment from the ground up
- Global enterprises often adopt a hybrid approach — EBR for strategic sites and ML automation for quick wins or selected processes
The right choice depends on your organisation's maturity, resources, timeline, and strategic priorities.
Recommended Digitalisation Roadmap
Automate Batch Review with ML Document Automation
Target the primary bottleneck first to achieve rapid efficiency gains with minimal disruption. Prove the value of digitalisation, structured data, and incremental change — before committing to larger investments.
Improve the Overall Process with Digital Workflows and Integrations
With digital review in place, assess opportunities to optimise the whole review process for maximum efficiency, speed, and quality. Build integrations into other systems such as QMS and SAP.
Expand into Digital BR and Full EBR
Deploy Paper-on-Glass based on the digitalised Master Batch Record. Over time, connect MES and laboratory systems for complete digitalisation and real-time automation.
💡 Key Insight
Software is a space where incremental learnings and roll-out tend to be more rewarding than rigid all-or-nothing implementations. Starting with ML automation builds internal confidence, proves the business case, and creates the structured data foundation every subsequent step depends on.
Conclusion: Balancing Speed, Flexibility, and Cost
There is no one-size-fits-all solution for digitalising batch record reviews. Each technology category comes with specific trade-offs between cost, complexity, and impact.
The key is to match technology choices to your organisation's specific needs, maturity level, budget, and strategic timeline. Software is a space where incremental learnings and roll-out tend to be more rewarding than rigid all-or-nothing implementations.
Start here → ML Review Automation
Fast to deploy, high ROI, no shop-floor disruption. The ideal first step for any organisation regardless of size.
Scale to → Full EBR & MES
Once the business case is proven and data foundations are in place, the path to full digitalisation is far smoother.
Not sure where your organisation sits on this map?
Talk to an Acodis expert — free 30-minute consultation. We'll assess your current process, identify the fastest path to ROI, and help you build a digitalisation roadmap that fits your budget and timeline.
Talk to an expert →


