If you've ever sat through a presentation about Electronic Batch Records (EBR) or Manufacturing Execution Systems (MES), you know the feeling. The promise is incredible: streamlined workflows, real-time data, paperless operations. Then you consider how to implement this within your premises and processes: 18 to 24 months timeline and an overall implementation cost that often exceeds $2 million for mid-size Pharma and CDMOs, excluding ongoing licence fees. Suddenly, that vision of digital transformation feels much further away.
Here's the reality: 60% to 70% of pharmaceutical companies and CDMOs are still doing batch record reviews the old-fashioned way, with paper documents or PDFs, manual checks, and hours of tedious verification work. Slow batch release, bored teams, errors missed. Not ideal, but it works and when the alternative requires such investments, it's hard to justify the leap.
What if there was a middle path? A way to start saving time and money now while gradually building toward full digitalization?
Enter Document Review Automation (DRA)
Thanks to advances in OCR and machine learning, there's a new option emerging that's changing how pharma companies think about batch record review digitalization. Document Review Automation (DRA) applied to Batch Records doesn't require you to rip and replace your entire system. Instead, it works with your current paper and PDF documents, automating the repetitive checks while building a foundation for future transformation. Turning complex documents with a lot of handwriting into structured information that computers can analyse.
Think of it this way: your batch records already contain structured logic. They have instructions, codified rules, signature requirements, date formats, calculations, and process steps that follow predictable patterns. All of these can be taught to a machine learning system - or better yet, the system can learn them through pattern recognition.
The beauty of DRA is that it doesn't try to replace human expertise. Quality experts are still essential for reviewing critical process parameters or steps, nuanced judgment calls and CAPAs. But they shouldn't be spending their valuable time checking hundreds of signatures and dates, verifying batch numbers, or catching basic calculation errors. That's where automation shines.
See batch record automation in action
Get a personalised demo and free digitalisation audit — no commitment required.
Book a free audit →The 60% Time Savings No One Talks About
Let's get specific about what this looks like in practice.
In a typical CDMO processing 2,000 batches per year, each batch record might be around 140 pages. Manufacturing spends about two hours reviewing each batch record, and QA spends around six hours. That's 16,000 hours annually just on batch record review—and that's before you factor in the back-and-forth when errors are caught late in the process, deeper analysis and remediation work.
With DRA, companies are seeing:
- 60% reduction in review time through automated standard checks
- 25% shorter batch release cycles by catching errors earlier in the process
- Faster inventory turnover because batches move through QA more quickly
- Better work satisfaction for operations and QA teams who can focus on meaningful analysis instead of box-checking and annoying corrections
Here's what makes it even more compelling: automated checks at the manufacturing stage (taking just 2-5 minutes) improve document quality before it reaches QA. This prevents the multi-day delays when QA sends documents back with questions. Avoiding just a few of those back-and-forth cycles can save entire weeks across your production schedule.
What You Actually See on Screen
So what does this look like day-to-day?
The system presents a visual interface that guides reviewers through all the automated checks and any detected errors. Instead of flipping through 140 pages looking for issues, reviewers see a curated list of items that need attention. They can:
- Approve or discard flagged items
- Leave comments for colleagues or audit trails
- Collaborate in real-time with team members
- Access historical data and patterns
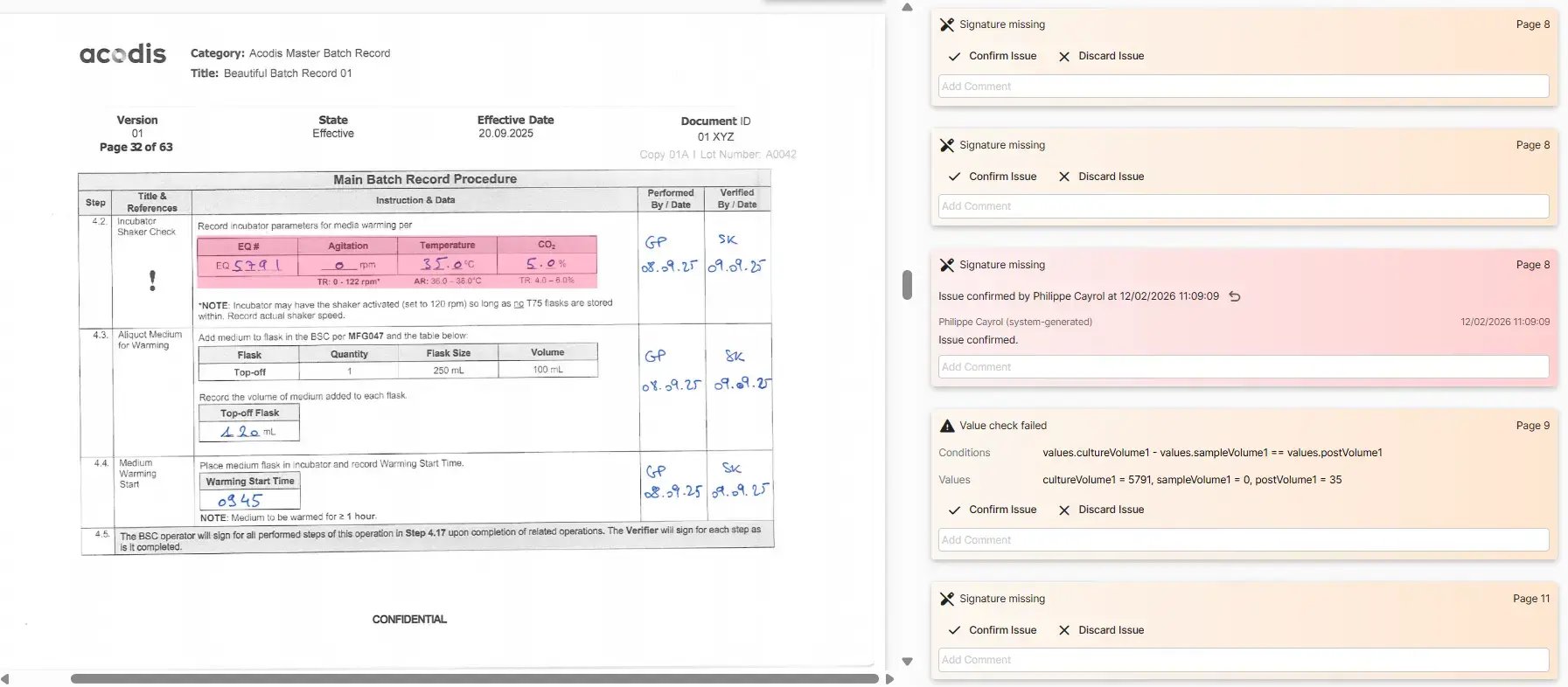
The platform tracks who reviewed what and when, creating a complete audit trail that actually improves compliance compared to paper-based processes.
Behind the scenes, the system is extracting structured data from your batch records—information about deviations, yields, process times, and more. This data becomes incredibly valuable for continuous improvement, trend analysis, and even regulatory submissions like Product Quality Reviews (PQRs).
The Path to Full EBR (When You're Ready)
Here's where it gets interesting for IT and digital transformation teams: DRA isn't just a stopgap solution. It's actually building the foundation for future digitalization.
As the system processes your batch records, it creates a digital library of all the rules, checks, and logic that underpin your Master Batch Records (MBR). Over time, this creates a smooth transition to "paper-on-glass"—digital forms that operators fill out on tablets, with immediate validation and standardized formats.
When you eventually implement connected production lines with machine sensors, that data can feed directly into your digital batch records, completing the journey to fully integrated EBR.
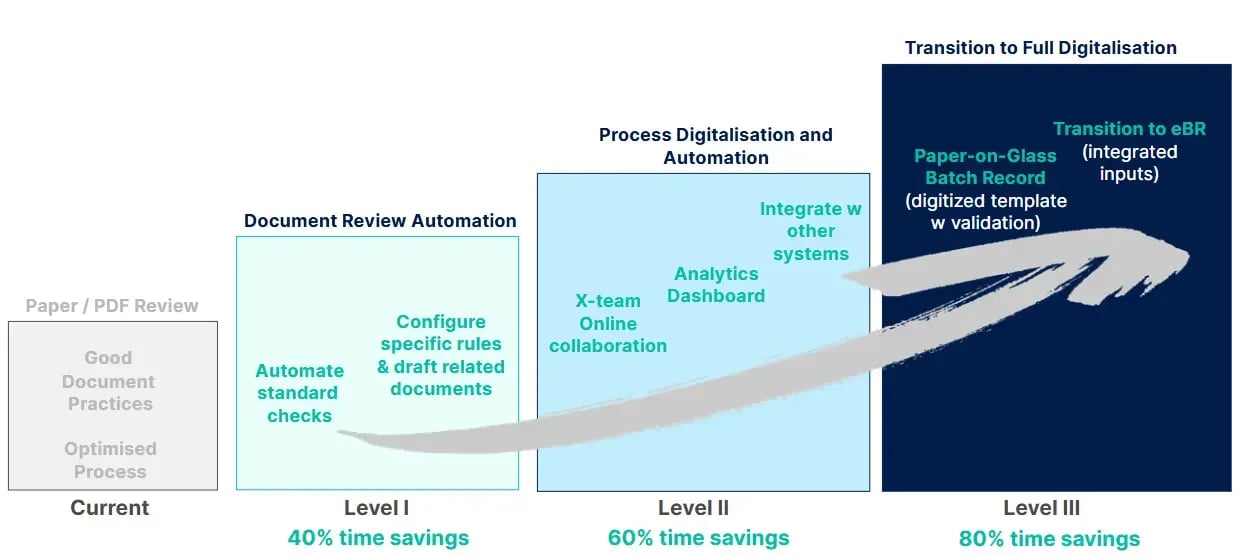
The digitalization journey looks like this:
- Level I (40% time savings): Automate standard checks on current PDF/paper documents, configure product-specific rules
- Level II (60% time savings): Add cross-team collaboration tools, analytics dashboards, and integration with other systems
- Level III (80% time savings): Transition to paper-on-glass with digital forms and eventually full EBR with integrated production data
The key difference? You can start at Level I in about three months and begin seeing ROI immediately, then progress to the next levels as your organization is ready.
The ROI Story That Gets Budget Approved
Let's talk numbers, because that's what gets digital transformation projects approved.
Illustrative ROI model — based on a mid-size CDMO processing 2,000 batches/year
Figures are representative estimates based on aggregated data from Acodis customer implementations. Actual results will vary.
Direct Savings
| 6,000 hours of review time saved annually | €540,000 |
| 2,000 hours saved on investigations & follow-up | €180,000 |
| 2,000 hours saved on internal document drafting | €180,000 |
Indirect Value
| Batch release time: 15 → 11 days (25% improvement) | €480,000 |
| Improved compliance (~1% of QA budget) | €280,000 |
| Total annual value | €1,660,000 |
| Average solution cost | €200,000/year |
| Annual ROI | 8.3x |
And here's the kicker: the implementation timeline is measured in months, not years. Initial assessment takes one month, roll-out to your first product takes two months, and scaling to all products typically happens within three months.
Why This Matters Now
The pharmaceutical industry is at an interesting inflection point. The pressure on efficiency and speed is intense, whilst the requirements on quality keep increasing. The technology for full digitalization exists and works - but the reality of implementation timelines, validation requirements, and capital constraints means most companies are stuck in a holding pattern.
Document Review Automation offers a pragmatic way forward. It delivers immediate value while building the infrastructure for future transformation. It proves the business case for digitalization with real ROI before asking for the big investment. And it allows teams to learn and adapt gradually rather than facing a big-bang implementation.
For QA managers tired of bottlenecks and repetitive work, it's a chance to show measurable improvement in cycle times and team satisfaction. For IT and digital transformation teams, it's a low-risk way to start the digitalization journey with a clear path to scale.
The question isn't whether to digitalize batch records—it's how to do it in a way that makes business sense for your organization today while setting you up for success tomorrow.
Getting Started
If this resonates with your current challenges, here are the practical next steps:
- Assess your current state: How many batches do you process annually? How long do reviews take? Where are the bottlenecks?
- Calculate your potential ROI: Use your actual numbers for batch volumes, review times, and current cycle times.
- Contact Acodis to get a free digitalisation audit and automation ROI analysis: we can guide you through the possibilities based on your specific circumstances
- Start with a pilot: Choose one product line to test the system and prove the concept.
- Plan your roadmap: Think about where you want to be in 12, 24, and 36 months—DRA can grow with you.
Ready to cut your batch review time in half?
Book a free 30-minute audit with our team. We'll map your current process, identify quick wins, and give you a tailored ROI estimate — no strings attached.
Book my free audit →


